Bentonite Clay Research Study: Lead from topically applied Bentonite clay concentrates in livers and kidneys of mice studied (March 2020, NIH: National Library of Medicine)
For those new to this website:
Tamara Rubin is a multiple-Federal-award-winning independent advocate for childhood Lead poisoning prevention and consumer goods safety, and a documentary filmmaker. She is also a mother of Lead-poisoned children (two of her sons were acutely Lead-poisoned in 2005). Since 2009, Tamara has been using XRF technology (a scientific method used by the U.S. Consumer Product Safety Commission) to test consumer goods for toxicants (specifically heavy metals — including Lead, Cadmium, Mercury, Antimony, and Arsenic). All test results reported on this website are science-based, accurate, and replicable. Items are tested multiple times to confirm the test results for each component tested. Tamara’s work was featured in Consumer Reports Magazine in February of 2023 (March 2023 print edition).
June 21, 2022 — Tuesday
Correction: The likely primary pathway for Lead exposure from bentonite clay sources noted in this particular study of mice was ingestion.
It is important to note — in reading this study — that this was not a study to determine the health and welfare (or potential for long-term chronic illness from Lead exposure) for the mice studied. This was a study simply to determine if Lead in clay might impact the outcome of other studies using mice IF the clay were used to soothe the skin of the mice — in response to lab activities that might cause skin issues during studies. The study’s conclusion was that BECAUSE the Lead from clay applied topically to the skin is then concentrated in the kidney and liver (likely primarily via ingestion, through grooming habits) this Lead exposure and accumulation might indeed compromise other scientific studies the mice are being used for and therefore the clay should be used with caution (if at all), when being used for this purpose.
I have included the abstract for the study, and the conclusion of the study below (both as an image and as a searchable text block). If you click on the image of the abstract it will take you to the full study — which is very interesting reading if you have the time. If for some reason in the future this link no longer works, please let me know — as I have also downloaded a full copy of the study to keep in my files). The three points below are the most important points relevant to human use of bentonite clay/ therapeutic clays.
Important points from the study linked and shown below are as follows:
- Samples of commercially available “therapeutic” bentonite clay/ green clays/ “healing” clays used in this study were found to have significant levels of Lead and Arsenic.
- In this study, mice treated with the Lead-contaminated-clay showed a statistically significant increase in the concentration of Lead in the livers and kidneys of the animals (compared to the control group).
- The most important takeaway from this — for people who might ingest or otherwise use bentonite clay — is this study demonstrating how this clay does not “detox” Lead (said another way: it does not remove Lead from the body), but instead clearly demonstrates that the Lead from Bentonite clay (or other “healing” clay sources) actually accumulates in the body in key organs (where it can cause health impairments later in life with chronic exposure).
Outside of the scope of this study, it has long been known that the impacts of Lead exposure are cumulative — and that long-term chronic Lead exposure can lead to life-threatening kidney and liver disease. We also know that Lead in the blood, bones, and organs can lead to many other chronic illnesses and health impairments (we don’t need this study to make any leap and draw that correlation for us — there is solid settled science that has already done that). (Link with more info on symptoms of Lead exposure in adults.)
- It is alarming that we — through this study — have clear evidence that Lead from a product applied to the surface of the skin (a product that may be accidentally ingested in much the same way as clay based toothpowder may be accidentally ingested by young children) is then concentrated in the liver and kidneys.
- For me, this study alone (as it relates to the known human impacts of even trace Lead exposure) provides enough evidence to advocate for the cessation of the use of bentonite clay in ANY potentially ingested applications (personal care products that may be accidentally ingested, toothpaste, supplements, etc.).
The image below is also linked to the original website with this study:
Text from the image above — bold sentences are of particular relevance:
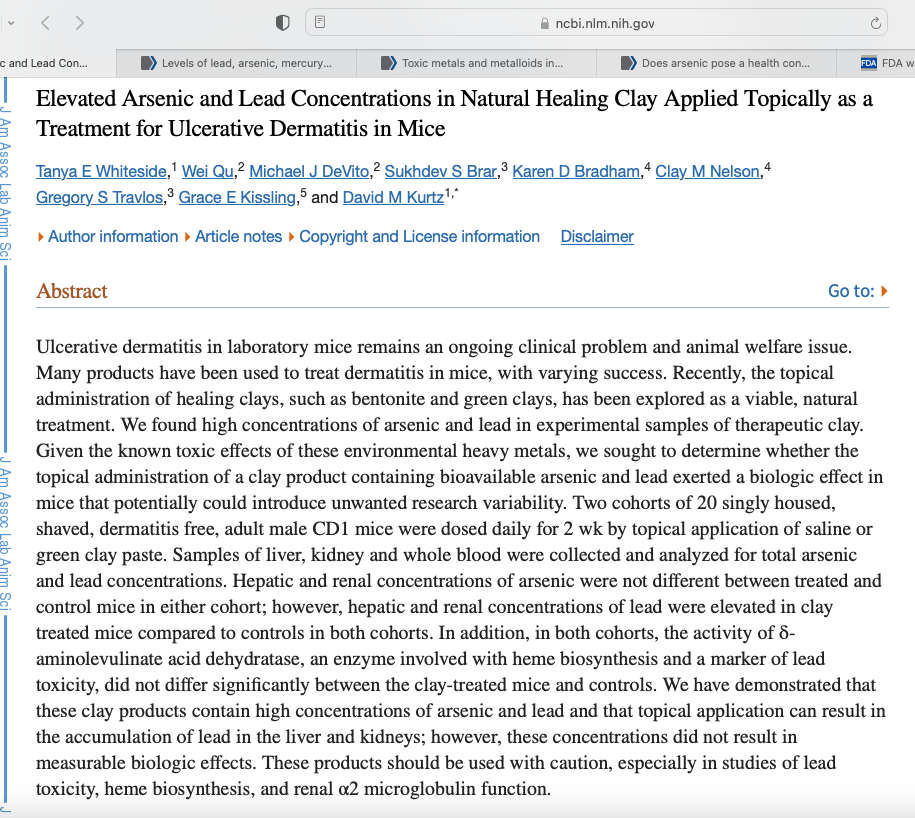
Abstract
Ulcerative dermatitis in laboratory mice remains an ongoing clinical problem and animal welfare issue. Many products have been used to treat dermatitis in mice, with varying success. Recently, the topical administration of healing clays, such as bentonite and green clays, has been explored as a viable, natural treatment. We found high concentrations of arsenic and lead in experimental samples of therapeutic clay. Given the known toxic effects of these environmental heavy metals, we sought to determine whether the topical administration of a clay product containing bioavailable arsenic and lead exerted a biologic effect in mice that potentially could introduce unwanted research variability. Two cohorts of 20 singly housed, shaved, dermatitis free, adult male CD1 mice were dosed daily for 2 wk by topical application of saline or green clay paste. Samples of liver, kidney and whole blood were collected and analyzed for total arsenic and lead concentrations. Hepatic and renal concentrations of arsenic were not different between treated and control mice in either cohort; however, hepatic and renal concentrations of lead were elevated in clay treated mice compared to controls in both cohorts. In addition, in both cohorts, the activity of δ-aminolevulinate acid dehydratase, an enzyme involved with heme biosynthesis and a marker of lead toxicity, did not differ significantly between the clay-treated mice and controls. We have demonstrated that these clay products contain high concentrations of arsenic and lead and that topical application can result in the accumulation of lead in the liver and kidneys; however, these concentrations did not result in measurable biologic effects. These products should be used with caution, especially in studies of lead toxicity, heme biosynthesis, and renal α2 microglobulin function.
Conclusion of this study (text below the image):
Text from the image above:
“In conclusion, we have demonstrated that 3 brands of commercially available healing clay products are naturally contaminated with high levels of arsenic and lead. Despite being unable to demonstrate any significant biologic effects, one brand that has the potential to cause harmful effects when entering the body showed a significant increase in the concentration of lead accumulation in the liver and kidneys of clay-treated mice compared with controls. For this reason, we discourage the use of natural healing clays in studies of lead toxicity, heme biosynthesis, or α2 microglobulin function. In other studies, the use of therapeutic clay products such as bentonite green clay should be considered carefully to minimize the potential for introducing unwanted variability.”
Never Miss an Important Article Again!
Join our Email List












Hi Tamara,
Just FYI, the links in the email around this article appear to be broken.
The page says: Error not found, 404 error
Thanks for the article, it’s been eye opening reads about “naturally safe” products that are not so safe at all!
Thank you! I will re-send that!
We add bentonite clay to our sons epsom salt baths as part of a detox regimen. Would you advise against this according to this research?
I’ve been looking into using bentonite clay as part of an armpit/lymphatic detox but now concerned that I should look for something else?
This is heartbreaking. A lot of tooth powders include bentonite clay because of its exfoliating benefits in removing plaque.
Bentonite clay is the primary ingredient of products like Pepto Bismol!
Clay has a looong tradition of skincare and digestive use in France. I really depended on it during my adult acne years; there is much literature.
I’m wondering if these heavy metals are in clay naturally or if the clay site was polluted before mining? With illegal dumping worldwide, that seems plausible.
It’s a complicated inquiry – and I don’t think it has been studied enough to determine safety in all applications (and until it has been, we need to be cautious as consumers.)
Tamara
What brands have you tested?
Greetings,
1. I would appreciate to receive information on the brands that were used in the study.
2,. Did you check where the clay from those brands came from? Did the clay come from areas that perhaps were already contaminated with lead and arsenic?
3. Who sponsored your research study?
4. Please share your opinion of how replicable your study is on mice compared to human beings, as they are from different species?
Thank you so much for your time
This is not my study – this is a research study linked in the NIH – click through to the study link and contact the researchers with these kinds of questions.