Lena’s Final Follow Up Post on My Page (so far)
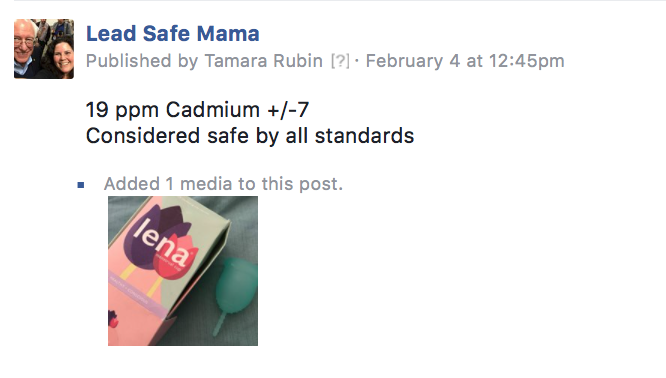 This is the final post from Lena on my Facebook Page. I included both a typed version (I typed it up, excuse any typos!) and a screenshot version here (grabbed from when they originally made the comment.) I can’t seem to find this comment in the hundreds of comments on my page. They may have deleted it or it may just be buried, but I thought it was important to have here on my site “for the record”! #MenstrualCupGate2018.
This is the final post from Lena on my Facebook Page. I included both a typed version (I typed it up, excuse any typos!) and a screenshot version here (grabbed from when they originally made the comment.) I can’t seem to find this comment in the hundreds of comments on my page. They may have deleted it or it may just be buried, but I thought it was important to have here on my site “for the record”! #MenstrualCupGate2018.
Note, the first image on this page (to your left if you are on a computer)is my original (unedited) post from when I initially shared the test results for their cups. My updated post can be seen in full here.
Click here to see the related posts
February 7, 2018
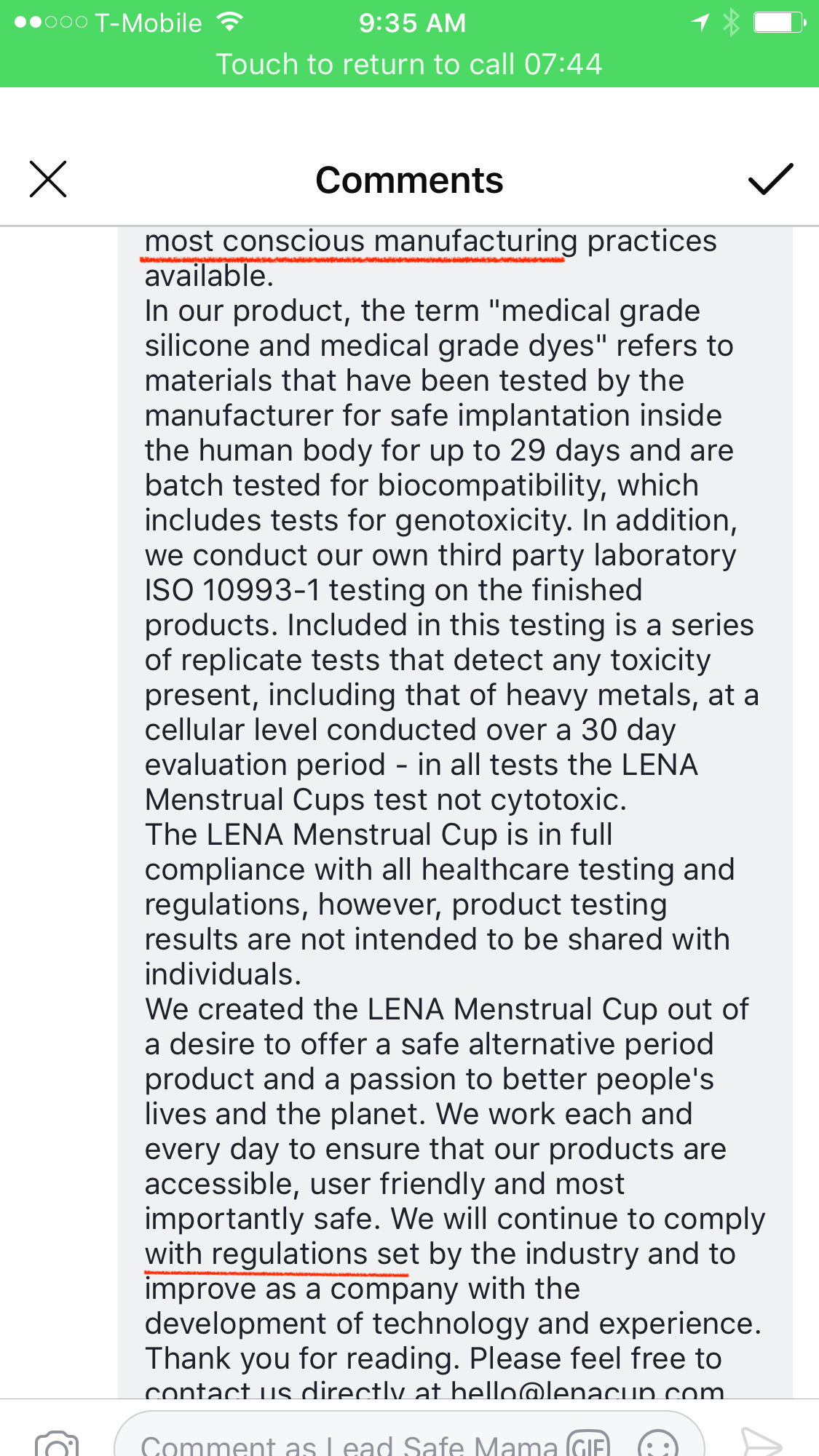
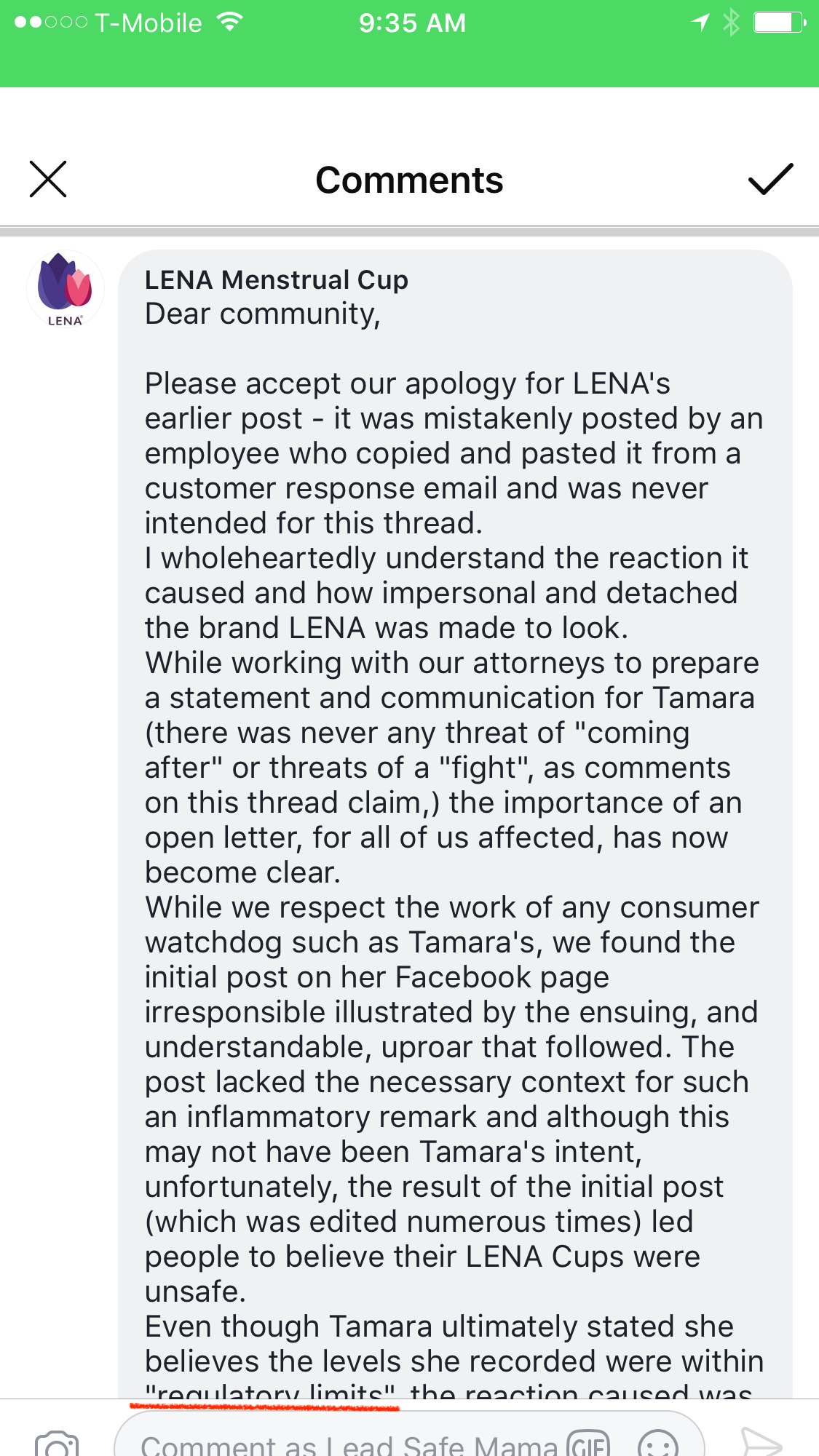
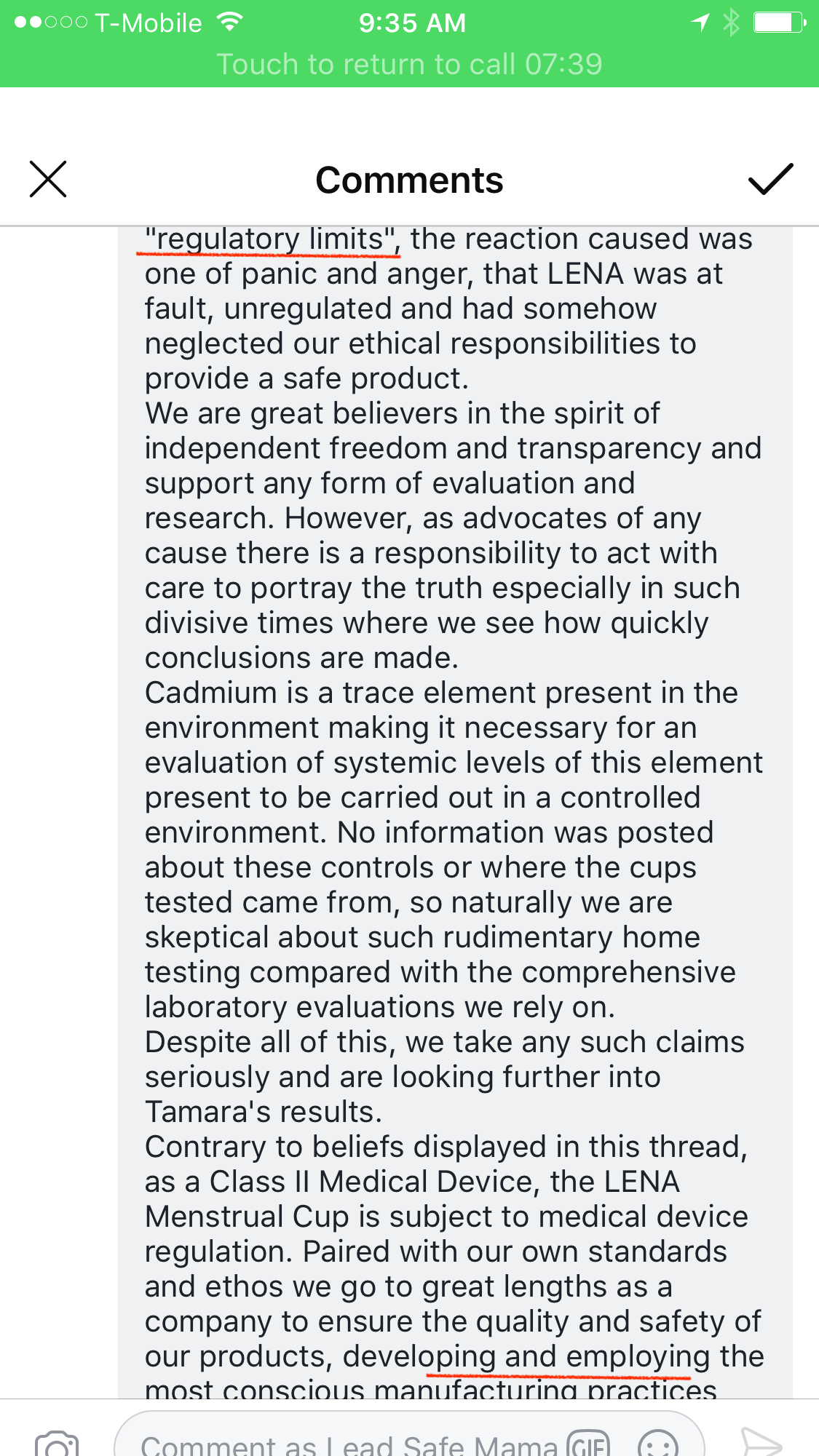
From: LENA Menstrual Cup
Dear community,
Please accept our apology for LENA’s earlier post – it was mistakenly posted by an employee who copied and pasted it from a customer response email and was never intended for this thread.
I wholeheartedly understand the reaction it caused and how impersonal and detached the brand LENA was made to look.
While working with our attorneys to prepare a statement and communication for Tamara (there was never any threat of “coming after” or threats of a “fight”, as comments on this thread claim,) the importance of an open letter, for all of us affected, has now become clear.
While we respect the work of any consumer watchdog such as Tamara’s, we found the initial post on her Facebook page irresponsible illustrated by the ensuing, and understandable uproar that followed. The post lacked the necessary context for such an inflammatory remark and although this may not have been Tamara’s intent, unfortunately, the result of the initial post (which was edited numerous times) led people to believe their LENA Cups were unsafe.
Even though Tamara ultimately stated she believes the levels she recorded were within “regulatory limits” the reaction caused was one of pain and anger, that LENA was at fault, unregulated and had somehow neglected our ethical responsibilities to provide a safe product.
We are great believers in the spirit of independent freedom and transparency and support any form of evaluation and research. However as advocates of any cause there is a responsibility to act with care to portray the truth especially in such divisive times where we see how quickly conclusions are made.
Cadmium is a trace element present in the environment making it necessary for an evaluation of systemic levels of this element present to be carried out in a controlled environment. No information was posted about these controls or where the cups tested came from, so naturally we are skeptical about such rudimentary home testing compared with the comprehensive laboratory evaluations we rely on.
Despite all of this, we take any such claims seriously and are looking further into Tamara’s results.
Contrary to beliefs displayed in this thread, as a Class II Medical Device, the LENA Menstrual Cup is subject to medical device regulation. Paired with our own standards and ethos we go to great lengths as a company to ensure the quality and safety of our products, developing and employing the most conscious manufacturing practices available.
In our product, the term “medical grade silicone and medical grade dyes” refers to materials that have been tested by the manufacturer for safe implantation inside the human body for up to 29 days and are batch tested for biocompatibility, which includes tests for genotoxicity. In addition, we conduct our own third party laboratory ISO 10993-1 testing on the finished products. Included in this testing is a series of replicate tests that detect any toxicity present, including that of heavy metals, at a cellular level conducted over a 30 day evaluation period – in all tests the LENA Menstrual Cup is in full compliance with all healthcare testing and regulations, however, product testing results are not intended to be shared with individuals.
We created the LENA Menstrual Cup out of a desire to offer a safe alternative period product and a passion to better people’s lives and the planet. We work each and every day to ensure that our products are accessible, user friendly and most importantly safe. We will continue to comply with regulations set by the industry and to improve as a company with the development of technology and experience.
Thank you for reading. Please feel free to contact us directly at hello@lenacup.com for any questions and comments.
Kindly,
Vili Petrova
Founder and President
Lena
Never Miss an Important Article Again!
Join our Email List



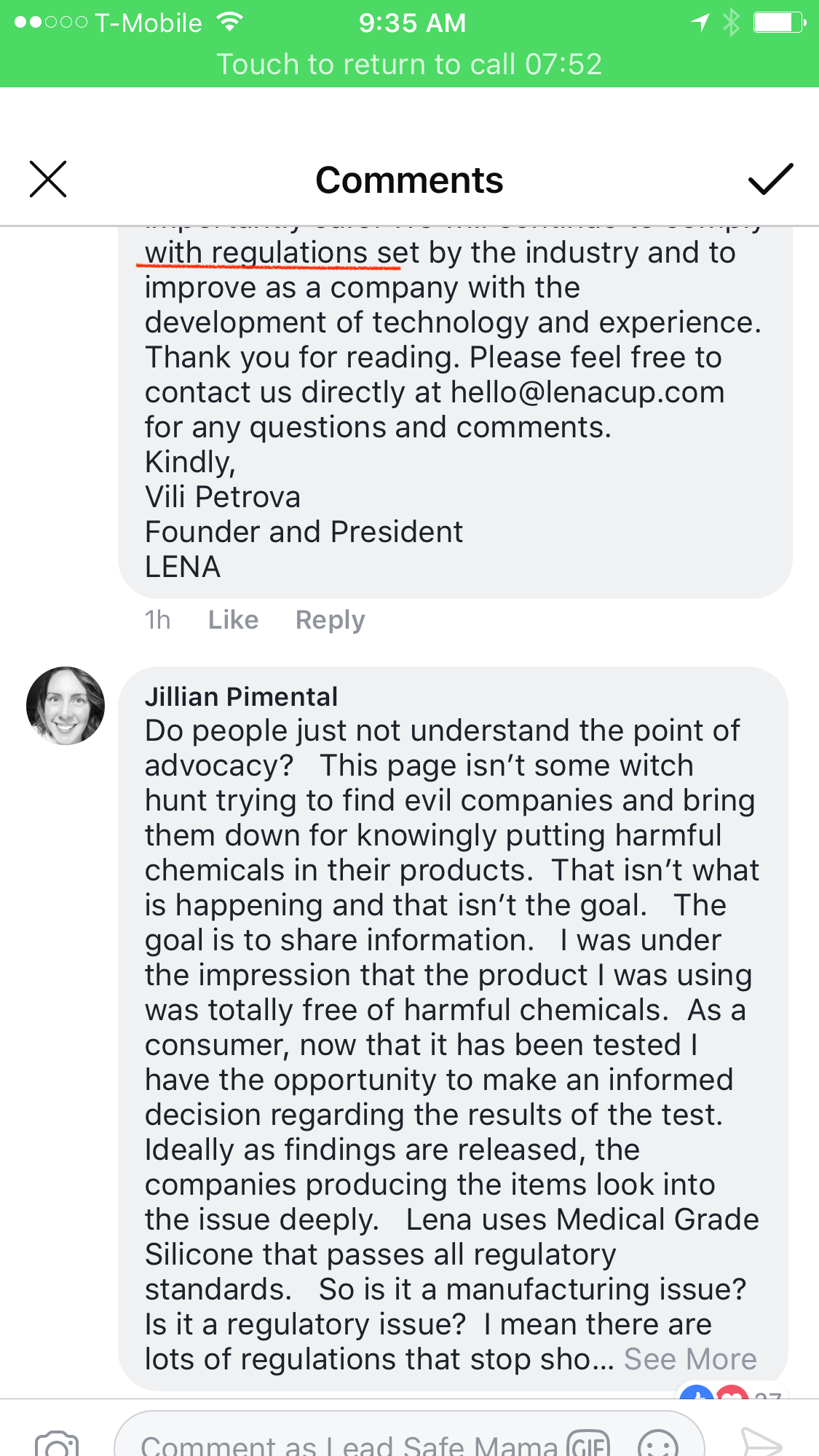

Where is the gif with the point flying over the stick figure’s head? There is so much to say, but at this point I feel much of it has already been said and they still have their cups shoved so far up their …ok, no, this comment isn’t going in a educational or neutral way, I apologize. I’ll step away and simmer for a bit. Seriously though, these gut reactions are getting people into trouble. I saw the original post (before edits) and read it as it being safe by the industry standards which is exactly what they follow. From there it is educational of ‘hey this is what is in it’. Aside from writing a short novel, there is no way to fully clarify what the intricacies of chemicals, testing, and regulations are! *leaving to simmer now*
Love
The menstrual cup brands are watching this and the Lena cup response is being tempered after the previous Diva cup official reply fiascoes. I’m guessing the other brands that you haven’t tested yet are already preparing their own “Thoughts and prayers” version of a reply on social media. Something along the lines of “Compliant and regulated manufacturing”, and the usual shade-throwing tactics:
1. “Tamara is using homemade testing/isn’t properly trained” >doubt her expertise and certifications. Make a personal attack.
2. “We are fully compliant” >insert ISO and technical jargon to sound good and deliberately browbeat upset consumers who aren’t scientists.
3. “This item is not from our facility” >trying to pretend it’s not theirs.
4. “But we are SO ecologically conscious!” >you’re still a business and you have quarterly targets to hit or exceed in sales. Related attacks: We are pro-women, therefore any bad news on our product means you’re anti-woman.
5. “Which trolls do we have on our payroll again?” >get a real person you secretly pay under the table because the 5-friend fake profile you created was quickly outed.
There is a pattern to your insinuative responses, menstrual cup manufacturers. Not everyone is dumb like you’re hoping. #wearewatchingyou
Thank you for writing this!
Wow, i just found your testing on menstrual cups! First, thank you Tamara for testing these!!
Next, I echo some of Angie’s thoughts – I’ll post some quotes from Lena, and then write my reaction.
1. “While working with our attorneys to prepare a statement and communication for Tamara (there was never any threat of “coming after” or threats of a “fight”, as comments on this thread claim,) the importance of an open letter, for all of us affected, has now become clear.”
I think the following quote, which was copied from their original response, shows that they were absolutely threatening Tamara with legal action. they were trying to walk that back, instead of apologizing to Tamara. It’s not against the law for anyone to test various aspects of a product and publish or share their findings online or wherever they choose. Isn’t this a free country? wow.
“we are consulting our attorneys regarding the distribution of false information and claims about our product without the support of scientific data and testing.”
2.While we respect the work of any consumer watchdog such as Tamara’s, we found the initial post on her Facebook page irresponsible illustrated by the ensuing, and understandable uproar that followed.”
How was Tamara’s original post irresponsible exactly? she stated that the level of cadmium found was within what is generally deemed as safe limits. their comment about this makes me so angry.
“The post lacked the necessary context for such an inflammatory remark and although this may not have been Tamara’s intent, unfortunately, the result of the initial post (which was edited numerous times) led people to believe their LENA Cups were unsafe.”
people will respond how they respond. Tamara’s post was truthful, simple, and factual. if people respond with dismay or anything else, that isn’t Tamara’s responsibility. We are all responsible for out own feelings. Also, why are they belittling all of these people’s dismay? they have a right to be dismayed. even if the level of cadmium is common in medical grade silicon products, people have a right to be dismayed about any level of heavy metals or cadmium. PLUS, Tamara has shown that not all silicon products test positive for cadmium, or even 22 ppm, as was the case with the Lena cup, so obviously, it is possible to make a silicon with lower levels than what was found in the Lena cup. so tired of companies being dismayed when consumer are dismayed about toxic substances, or dismayed when consumers get proactive and start testing for truthfulness of advertising claims. also, maybe companies should take the responsibility of being completely transparent – and make testing results available to consumers, even if levels of cadmium are below the “safe limit.” then, there won’t BE any “uproar”. they could invite other silicon manufacturers to also be transparent, and they could win the trust of consumers this way.
3. “Even though Tamara ultimately stated she believes the levels she recorded were within “regulatory limits” the reaction caused was one of pain and anger, that LENA was at fault, unregulated and had somehow neglected our ethical responsibilities to provide a safe product.”
FALSE. again, Tamara stated that the level of cadmium was considered “safe” by all standards. Why do they repeatedly project their own fears onto Tamara? makes me so angry.
“We are great believers in the spirit of independent freedom and transparency and support any form of evaluation and research. However as advocates of any cause there is a responsibility to act with care to portray the truth especially in such divisive times where we see how quickly conclusions are made.”
It doesn’t sound like they are believes in independent freedom and transparency. if they are transparent, post your own testing results for all to see from the beginning of selling a product. Tamara said nothing inflammatory in her original posting of her testing results.
4. “No information was posted about these controls or where the cups tested came from, so naturally we are skeptical about such rudimentary home testing compared with the comprehensive laboratory evaluations we rely on.” “…Cadmium is a trace element present in the environment making it necessary for an evaluation of systemic levels of this element present to be carried out in a controlled environment. No information was posted about these controls or where the cups tested came from, so naturally we are skeptical about such rudimentary home testing compared with the comprehensive laboratory evaluations we rely on.”
Tamara does post about how she tests items, and they could have asked her for even more detail on how she tests. Clearly their product contains cadmium, as other silicon products Tamara has tested i her home contained a variety of levels of cadmium – some less, some more than the Lena cup, i think. I get what they are saying — that a lab is “completely sterile,” but actually, we as consumers don’t know that — we’re not there. the time of trusting the ‘expert’ is somewhat over. consumers have been duped too many times. i for one am willing to believe Tamara’s results just as much as the results coming out of a “sterile” lab. it’s a professional XRF instrument for goodness sake. the only thing that could be done to make all of this more sure is to test the cadmium levels of the air of dust of Tamara’s home at a variety of points throughout the year. Obviously, that would cost a lot of money. I don’t see any company, including Lena, doing testing with an XRF instrument AND MAKING THE RESULTS TRANSPARENTLY AVAILABLE, for consumers.
5. “Despite all of this, we take any such claims seriously and are looking further into Tamara’s results.” I wish they could say this one somewhat positive thing without being condescending.
6. “have been tested by the manufacturer for safe implantation inside the human body for up to 29 days and are batch tested for biocompatibility, which includes tests for genotoxicity. In addition, we conduct our own third party laboratory ISO 10993-1 testing on the finished products. Included in this testing is a series of replicate tests that detect any toxicity present, including that of heavy metals, at a cellular level conducted over a 30 day evaluation period – in all tests the LENA Menstrual Cup is in full compliance with all healthcare testing and regulations, however, product testing results are not intended to be shared with individuals.”
I don’t know about you, but I could easily wear my menstrual cup for 6 days each month, which means that I’ve already chalked up 29 days of wearing the cup after only a little less than 5 months. Since I started wearing a menstrual cup, I’ve probably worn it for at least 720 days in the last 10 years. I understand that it would be hard to do testing for 720 days, or even half that, but this simply demonstrates that testing always falls short of real use scenario, so, although their test of 29 or 30 days is good and I’m glad they did it, we still have the right to be concerned about ANY level of toxic substances found in the product. yeah, and i have no idea what ISO 10993 is….it would be better to spend time writing up a post about what that is, so we can decide for ourselves if it means anything to us as consumers. And why, why, is “product testing results [not[ intended to be shared with individuals” ???!!?!?
with almost every technological advancement, we find later unintended consequences. the menstrual cup has made a positive impact on my lifestyle, but maybe it is having unintended consequences on my health or fertility, regarding any level of toxicity found in such a product. i’d like to know about any level, even if its within the “safe limit” so i can decide for myself if I want to partake. We should have that right to know all information, and decide for ourselves…i don’t want the ISO 10993 to decide for me.
now to read the results from the other menstrual cups tests….i just had to respond with some of my thoughts before moving on because it help me overcome the anger I feel that people or companies with lots of money or assumed power try to persecute those who are simply on a quest for PRECISE and full transparency truth, as is Tamara. She is doing the work we all wish we could do in our homes, and is sharing with all.
thank you Tamara.
🙂
Thank you for taking the time!